Time is Limited - Act Now

Cartiva® implant details: The background and development of a harmful medical procedure
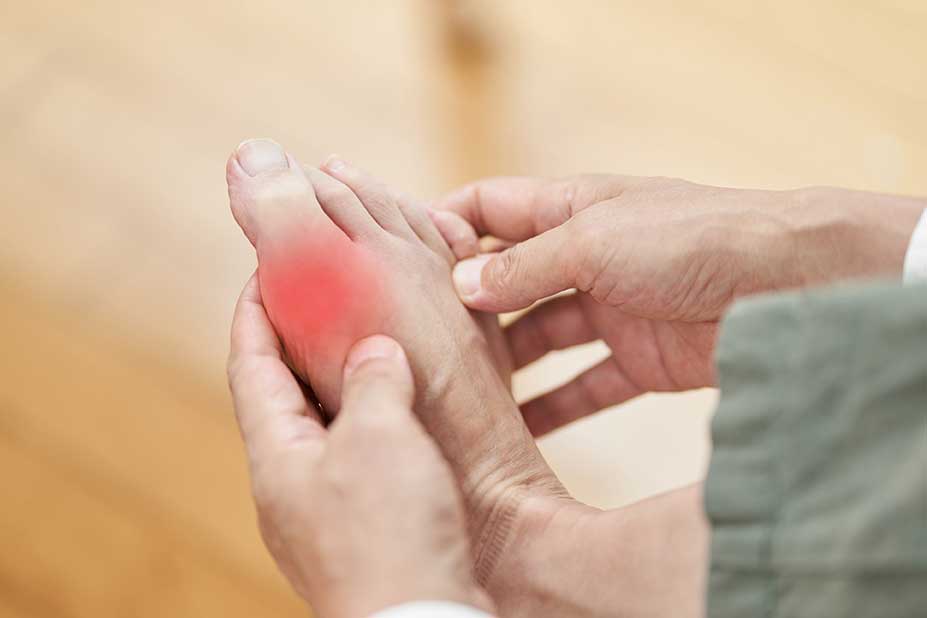
The FDA approved the Cartiva® Synthetic Cartilage Implant (SCI) in July 2016 for treating mid-stage to advanced arthritis at the base of the great toe.
In 2018, Wright Medical paid an estimated $435 million to acquire Cartiva®.
The Cartiva® implant is made from saline solution and polyvinyl alcohol–the same material as in soft contact lenses–and is almost the same size and shape of a mini-marshmallow.
The company has touted a variety of benefits that include:
- The reduction of joint pain without sacrificing the foot's natural range of motion;
- The retention of the patient's mobility and movement; also,
- Patients may return to daily activities faster than patients who undergo a traditional fusion procedure, because of a less restrictive rehabilitation protocol.
Composed of a biocompatible, durable, low-friction organic polymer, the Cartiva® implant is designed to function similarly to natural cartilage as it compresses and expands much like real cartilage–and it can be implanted in about 35 minutes.
Cartiva® implant dangers: What makes this dangerous medical device an injury waiting to happen
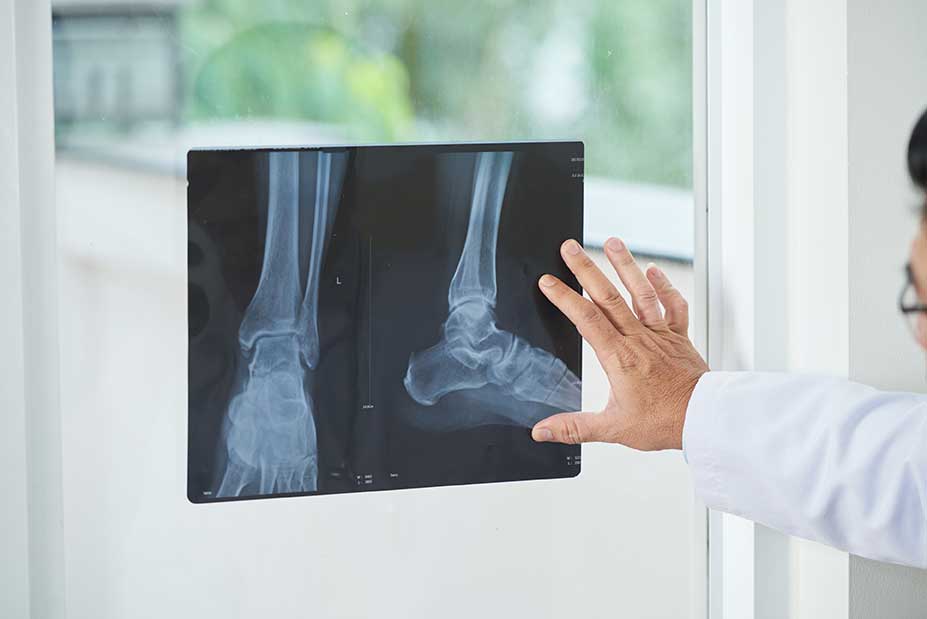
Known as the MOTION trial, Cartiva®'s pivotal trial showed significantly higher pain scores, including a case series with mixed results and numerous online complaints from dissatisfied implant patients.
The following elements make the Cartiva® medical implant a potential danger to patients:
- Smooth implant surface. Because the Cartiva® implant is very smooth on all surfaces, it creates a functional flaw in the design that may lead to serious injuries post-operation.
- Subchondral plate violation. Without subchondral plate support–and with no alternative means of support–the Cartiva® implant situates into the soft metaphyseal bone, making it prone to impaction failure.
- Little to no osseointegration. Osseointegration, or ingrowth of bone into the implant, is described as an essential aspect of implant stability–the absence of which is detrimental to surgical success
- Placement in soft bone offers little support. Implant of Cartiva® into the supporting bone means the structure is too weak to provide the meaningful, long-term support required to resist the load applied to the implant.
“While some of the patients are satisfied with Cartiva®, a majority are dissatisfied with many citing pain that lasts longer than they expected," reveals Mike Matson, a medtech analyst at Needham & Co.
The Shield Justice Watch team believes that patients that received a Cartiva® implant and then suffered a serious medical injury diagnosis should be able to seek justice and compensation from the manufacturer in a court of law.
Our network of attorneys are ready to support you.
IS THERE A CASE?
Answer a brief, private online questionnaire that asks for key details of the experience
EXPERIENCED LAWYERS
Have the case evaluated by a legal team free of charge without any obligation to file
GET STARTED NOW
Depending on the circumstances, the case could be filed for potential compensation
Please seek the advice of a medical professional before making health care decisions. This advertisement is not associated with Cartiva® or any government agency.
www.shieldjusticewatch.com is the property of Shield Legal LLC. 7180 Pollock Drive, 2nd Floor, Las Vegas, NV 89119
This website is not part of the Facebook website or Facebook, Inc. Additionally, this site is NOT endorsed by Facebook in any way. FACEBOOK is a trademark of FACEBOOK, INC.
ATTORNEY ADVERTISING. This Website is not intended to provide medical advice. Consult your doctor or physician before starting or stopping any medication.
Discontinuing a prescribed medication without your doctor’s advice can result in injury or death. are not an indication of future results. Every case is evaluated on its own facts and circumstances. Valuation depends on facts, injuries, jurisdiction, venue, witnesses, parties, and testimony, among other factors. No representation is made that the quality of legal services to be performed is greater than the quality of legal services performed by other lawyers. Shield Justice Watch does not itself provide legal services. Cases will be referred to third party attorneys and law firms. Do not rely on this advertisement in making any medical decision. Please call your physician before making any medical decision, including altering your use of any drug. Court costs and case expenses may be the responsibility of the client. Not available in all states. This advertisement is not intended as a testimonial, endorsement or dramatization, and does not constitute a guarantee, warranty, or prediction regarding the outcome of your legal matter, either expressed or implied. Anyone considering a lawyer should independently investigate the lawyers' credentials and ability, and not rely upon advertisements or self-proclaimed expertise. Only persons age 18 or older have permission to access our Service. Our Service does not address anyone under the age of 13("Children").
Privacy Policy | Terms and Conditions | CCPA Privacy Notice | Do Not Sell My Info
©2024 Shield Justice Watch. All Rights Reserved